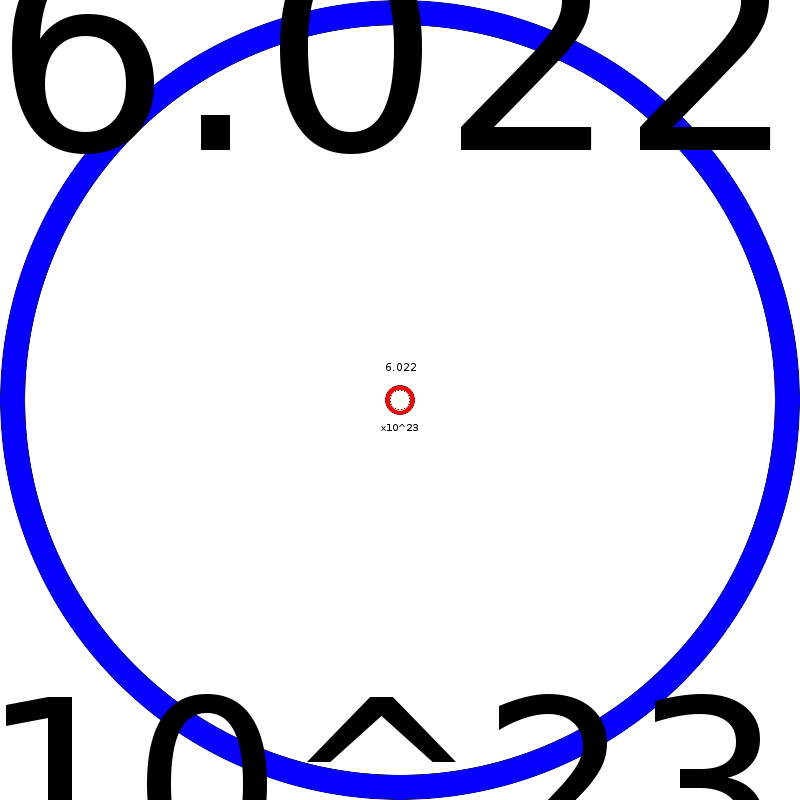
| Abstraction is stripping away the peripherals of a particular reality to get at the essence of its being. For my abstraction, I chose three representations: two visual and one textual representation of the mole(although the textual representation has visual aspects to it as well). One of the key concepts of the mole is that it is both an incredibly large number and while at the same time, the atom is an incredibly small piece of matter. For figure 1, I started with a very small dot that is hard to see (like an atom) and copied it over and over again until I got a very large number that would be incredibly difficult to count. While it isn't exactly the number that a mole represents, it gets at the essence of what a mole is – a large, uncountable number – and in chemistry we count an incredibly small object – the atom. For figure 2, I contrasted a large circle with a small circle – highlighting the essence of enormity of the number (too big to fit on the paper) and the minuscule nature of what the number is counting (too small to read). For figure 3, I contrast again the large and small principle, but using words that get incredibly small and incredibly large – it forces one to extrapolate what the rest of the sentence would say. Like many abstractions, not all of the picture is obvious at the beginning, but can be inferred and interpreted. These abstractions hopefully can help students capture the essence of what I'm trying to teach them about the mole and the implications for chemistry. The enormity and the tininess of the mole is an important dichotomy that can't be emphasized enough and hopefully abstracting it down can help students grasp the concept. |
|
0 Comments
 A pattern is a repeating sequence of a common variable often in an expected manner, but sometimes unexpected. In dealing with the mole in chemistry, we often have many calculations – calculating the molar mass of a substance, converting between mass and moles, and calculating the heat of enthalpy of a reaction – just to name a few. For this exercise of patterning, I will use the calculation of molar mass as my example. Calculating the molar mass of a substance follows a particular pattern or method. Once they students learn the pattern of calculation, most of my students report enjoying this type of chemistry calculation. As indicated by the diagram below, you can calculate the molar mass of any substance as long as you know the composition of elements. The pattern is the following: multiply the number behind the element symbol (the subscript) by the atomic mass from the periodic table. Do that for each element given for the compound then add up all of the masses together. Written out... Atomic mass x number + atomic mass x number + atomic mass x number …. etc... The pattern that some students have difficulty learning is when to multiply and when to add. Sometimes they do the opposite order or they may simply multiply all the numbers together. In brainstorming for new patterns with this calculation, I noticed that we always use the addition symbol (+) between different elements. I've never specifically told the students that connecting pattern. When drawing out the calculation, it looked like a cross or a bridge – perhaps a bridge from one element to another! This could be an important memory cue for students to remember to use addition when going between elements. Switching to a different element, they need to make a bridge first! That's a pattern they can remember every time they do a molar mass calculation.
Interview with Tom Kapanka, administrator of Calvary Christian Schools, a K-12 school
|
Jonathan Langdon
Technology Facilitator at Calvary Christian Schools. High School Science Teacher since 2008. CC License
This work is licensed under a Creative Commons Attribution 4.0 International License. Archives
February 2015
Categories |

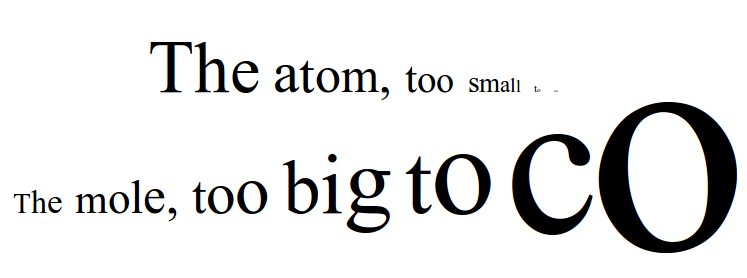

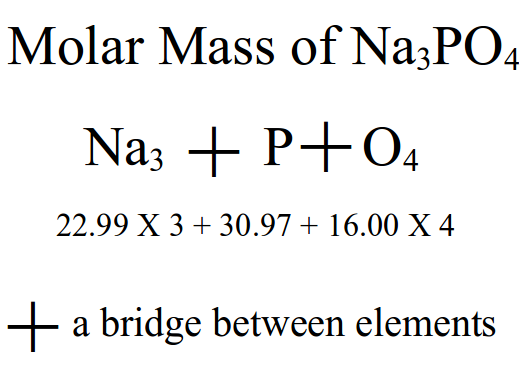

 RSS Feed
RSS Feed
